Specification of health data transfer from devices to DiGA (§ 374a SGB V)
Seiteninhalt:
Medical aids and implants often can measure various kinds of data, depending from the kind of device and the concrete product. Not all of this data can be considered by mid 2027 when § 374a SGB V comes into effect. Therefore an assessment on typical use cases within prioritized domains was conducted in order to discover the kinds of data that are especially relevant for care scenarios which can be supported by DiGA (see chapter on Methodology).
This page lists these Mandatory Interoperable Values (MIVs) for the domains that have been assessed so far. Each MIV is described by a structured profile in table form that contains all relevant references for implementers of Device Data Recorders and DiGA.:
| Defining ValueSet | Each MIV is defined by a FHIR ValueSet which is referenced in the MIV profile as the defining value set. This ValueSet contains the LOINC codes for the data that implement the MIV. In order to implement the MIV, a DiGA MUST be able to process at least one of these LOINC codes. In order to implement the MIV, a Device Data Recorder MUST be able to provide data that complies at least to one of these LOINC codes. |
| SMART Scopes | DiGA are only allowed to retrieve data elements explicitly covered by the granted SMART scopes. These scopes are defined per MIV and listed here. |
| FHIR Observation Profile | Device Data Recorders provide measured data as FHIR Observation resources. Each MIV is bound to one or more FHIR resource profiles that define the structure and semantics of the provided resources. The Observation profiles that are defined for the specific MIV are listed in this row of the MIV profile table. |
| FHIR Interactions | Access to the resources is given through standard FHIR read and search RESTful interactions and - if the MIV covers aggregated data - FHIR operations. These interactions and operations are described per MIV including MIV-specific examples. A reference to this description is given here. |
| Aggregated Data | Data from a MIV’s measured data may be aggregated as a standardized report. If such a report is defined for a specific MIV, a reference to the respective FHIR specification is given here. |
| Vendor Holdup | The Vendor Holdup defines the maximum acceptable delay between data availability in the Health Record and data availability through the HDDT interface. The Vendor Holdup is given in seconds delay and defined per MIV based on the MIV-specific use cases. E.g. if a Vendor Holdup of 60 seconds is defined for a MIV, the Device Data Recorder MUST be able to provide the measured data to a DiGA at last 60 seconds after he received that data from the Personal Health Device through the device’s Personal Health Gateway. |
Historic-Data-Period |
The Historic Data Period defines the period for which a Device Data Recorder MUST be able to provide data back into the past. The Historic Data Period is defined by the Device Data Recorder per MIV. A DiGA can retrive the specific value for a given Device Data Recorder and MIV through the BfArM HIIS-VZ API. This row of the MIV profile defines the minimum acceptable value for the Historic Data Period for the given MIV. E.g. if a Historic Data Period of 30 days is given in the MIV profile, a Device Data Recorder MUST be able to serve a request for data that was measured 30 days ago. Nevertheless, a Device Data Recorder can define its MIV-specific Historic Data Period to 60 days to signal connecting DiGA that they may even obtain data which is 7 weeks old. But in the given example, the Device Data Recorder would not be allowed to set its Historic Data Period value to 14 days, because the acceptable minimum is defined as 30 days for the MIV. |
Grace-Period |
A DiGA may request data from a Device Data Recorder at any time. However, to avoid overload situations on the Device Data Recorder side, a Grace Period is defined per MIV. This Grace Period defines the minimum time span that a DiGA MUST wait after a previous request for data from the same patient before a new request for data from that patient can be sent to the Device Data Recorder. If a DiGA sends a request for data from a patient before the Grace Period has elapsed since the last request for that patient, the Device Data Recorder MAY reject the new request. The Grace Period is defined by the Device Data Recorder per MIV. A DiGA can retrive the specific value for a given Device Data Recorder and MIV through the BfArM HIIS-VZ API. This row of the MIV profile defines the maximum acceptable value for the Grace Period for the given MIV. E.g. if a Grace Period of 60 minutes is given in the MIV profile, a Device Data Recorder MUST be able to serve requests from a DiGA for a certain patient’s data with a frequency of one request per hour. Nevertheless, a Device Data Recorder can define its MIV-specific Grace Period to a lower value, e.g. 10 minutes in this example, to signal connecting DiGA that they may even obtain data with higher frequency. But in the given example, the Device Data Recorder would not be allowed to set its Grace Period value to 120 minutes, because the acceptable maximum is defined as 60 minutes for the MIV. |
Self-management tools used by patients to determine glucose levels analyze either capillary blood (“bloody measurement” on the fingertip) or interstitial fluid (e.g. measurements performed through real-time Continuous Glucose Monitoring (rtCGM)). Other measurement methods - e.g. using arterial blood - are primarily used in the doctor’s office and are not taken into account for the HDDT Usecase “Diabetes Self-Management”.
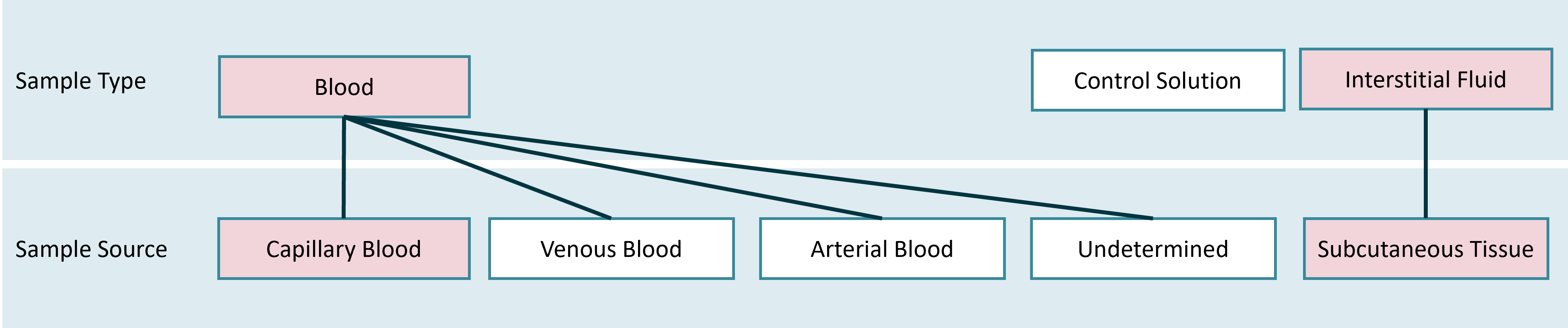
For the initial specification (version 1) of HDDT, two mandatory interoperable values (MIVs) are defined for diabetes self-management:
The table below lists typical use cases and certification relevant systems for these MIVs.
| MIV | examples of typical use cases | examples of certification relevant systems |
|---|---|---|
| Blood Glucose Measurement | • monitoring of patients with a combination of basal insulin with oral antidiabetic drugs • diabetes diary for DMT2 patients |
• blood glucose meter (glucometer) |
| Continuous Glucose Measurement | • supporting patients in flattening glucose curves • analyzing behavioral effects on glucose level • gathering clinical metrics for measuring the status of the diabetes therapy • analyzing hidden hypoglycaemias |
• real-time Continuous Glucose Monitoring (rtCGM) • Closed Loop Systems |
The MIV Blood Glucose Measurement covers values from “bloody measurements” using capillary blood from the finger tip. Measurements are performed based on a care plan (e.g. measuring blood sugar before each meal) or ad hoc (e.g. a patient feeling dim what may be an indicator for a hypoglycamia). Values are very accurate and therefore best suited for therapeutical decision making.
| Defining ValueSet | The MIV Blood Glucose Measurement is defined by the FHIR ValueSet Blood Glucose Measurement from LOINC. This ValueSet contains LOINC codes for blood glucose measurements using blood or plasma as reference methods with the values provided as mass/volume and moles/volume. In addition more granular LOINC codes for “Glucose in Capillary blood by Glucometer” provided as mass/volume and moles/volume are included with the value set because these codes are already in use by several manufacturers of glucometers. |
| SMART Scopes | patient/Observation.rs?code:in=https://gematik.de/fhir/hddt/ValueSet/hddt-miv-blood-glucose-measurement patient/Device.rs patient/DeviceMetric.rs |
| FHIR Observation Profile | Device Data Recorders provide Blood Glucose Measurement to DiGA using the MIV-specific Observation Profile HDDT Blood Glucose Measurement. This profile allows to capture a single blood glucose value as a FHIR Observation resource. |
| FHIR Interactions | Access to the resources is given through standard FHIR read and search RESTful interactions as described in the MIV-specific API. |
| Aggregated Data | By now there are no aggregated data defined for the MIV “Blood Glucose Measurement”. |
| Vendor Holdup | The maximum acceptable delay between data availability in the Health Record and data availability through the HDDT interface is 60 seconds. |
Historic-Data-Period |
Device Data Recorders that provide Blood Glucose Measurement data MUST make the measured values retrievable for at least 30 days. |
Grace-Period |
A Device Data Recorder MAY reject a DiGA’s request for a patient’s Blood Glucose Measurement if the previous request for that patient was answered less than 15 minutes ago. |
The MIV Continuous Glucose Measurement covers values from continuous monitoring of the glucose level, e.g. by rtCGM in interstitial fluid (ISF). Measurements are performed through sensors with a sample rate of up to one value per minute. By this Continuous Glucose Measurement can e.g. be used to assess dependencies between a patient’s individual habits and behaviours and his glucose level. Due to the high density of values over a long period of time, many clinical metrics can be calculated from Continuous Glucose Measurement which help the patient and his doctor to easily capture the status of the patient’s health and therapy.
| Defining ValueSet | The MIV Continuous Glucose Measurement is defined by the FHIR ValueSet Continuous Glucose Measurement from LOINC. This ValueSet includes codes relevant to continuous glucose monitoring (CGM) of ISF glucose, considering mass/volume and moles/volume as commonly used units. In the future codes defining non-invasive glucose measuring methods may be added to this value set. |
| SMART Scopes | patient/Observation.rs?code:in=https://gematik.de/fhir/hddt/ValueSet/hddt-miv-continuous-glucose-measurement patient/Device.rs patient/DeviceMetric.rs |
| FHIR Observation Profile | Device Data Recorders provide Continuous Glucose Measurement to DiGA using the MIV-specific Observation Profile HDDT Continuous Glucose Measurement. This profile allows to share sampled glucose values as FHIR Observation resources. Each resource holds multiple values while the time stamp of each value can be determined by the time stamp of the first value and the fixed sample rate. |
| FHIR Interactions | Access to the resources is given through standard FHIR read and search RESTful interactions as described in the MIV-specific API. |
| Aggregated Data | As stated above, Continuous Glucose Measurement sampled data are a basis for many clinical metrics used in diabetes therapy monitoring, e.g. times in ranges (e.g. times in hypoglycemia and hyperglycemia) and Glucose Management Index (GMI). Device Data Recorders MUST provide the structured, coded part of the HL7 CGM Summary Report to DiGA using the MIV-specific Profile HDDT CGM Summary Report. This profile allows to share a well defined set of relevant clinical metrics as FHIR Observation resources. Access to the summary report is provided through the FHIR operation $hddt-cgm-summary. |
| Vendor Holdup | The maximum acceptable delay between data availability in the Health Record and data availability through the HDDT interface is 15 minutes. |
Historic-Data-Period |
Device Data Recorders that provide Continuous Glucose Measurement data MUST make the measured values and aggregated clinical metrics (per CGM Summary Report) retrievable for at least 30 days. |
Grace-Period |
A Device Data Recorder MAY reject a DiGA’s request for a patient’s Continuous Glucose Measurement if the previous request for that patient was answered less than 15 minutes ago. |
In Germany, more than ten million people live with chronic lung and respiratory diseases, which significantly affect their quality of life. The most common conditions include bronchial asthma, chronic obstructive pulmonary disease (COPD), and obstructive sleep apnea syndrome (OSAS or simply sleep apnea). These conditions often require continuous monitoring and therapy. This is where aids such as peak flow meters for lung function measurement come in, allowing those affected to have more control over their condition and improving the quality of treatment.
For the initial specification (version 1) of HDDT, one mandatory interoperable value (MIV) is defined for lung function testing:
The table below lists typical use cases and certification relevant systems for this MIV:
| MIV | examples of typical use cases | examples of certification relevant systems |
|---|---|---|
| Lung Function Testing | • DiGA to support treatment of asthma bronciale • DiGA for compliance monitoring |
• peak flow meter • spirometer |
The MIV Lung Function Testing covers values from measurements using peak flow meters or spirometers to monitor lung function in patients with (chronic) respiratory diseases. Measurements are performed based on a care plan (e.g. measuring peak expiratory flow (PEF) in the morning and evening) or ad hoc (e.g. a patient experiencing breathing difficulties).
| Defining ValueSet | The MIV Lung Function Testing is defined by the FHIR ValueSet Lung Function Testing. This ValueSet contains LOINC codes for individual lung function testings (PEF and FEV1), LOINC codes for associated lung function reference values (e.g. personal best), and LOINC codes for relative lung function values, calculated in percentages (e.g. PEF relative to a reference value). |
| SMART Scopes | patient/Observation.rs?code:in=https://gematik.de/fhir/hddt/ValueSet/hddt-miv-lung-function-testing patient/Device.rs patient/DeviceMetric.rs |
| FHIR Observation Profile | Device Data Recorders provide Lung Function Testing related measurements and derived values to DiGA using the MIV-specific Observation Profiles HDDT Lung Function Testing, HDDT Lung Function Reference Value, and HDDT Lung Function Testing Complete. |
| FHIR Interactions | Access to these resources is given through standard FHIR read and search RESTful interactions as described in the MIV-specific API. |
| Aggregated Data | By now there are no aggregated reports defined for the MIV “Lung Function Testing”. |
| Vendor Holdup | The maximum acceptable delay between data availability in the Health Record and data availability through the HDDT interface is 60 seconds. |
Historic-Data-Period |
Device Data Recorders that provide Lung Function Testing data MUST make the measured values retrievable for at least 14 days. |
Grace-Period |
A Device Data Recorder MAY reject a DiGA’s request for a patient’s Lung Function Testing data if the previous request for that patient was answered less than 15 minutes ago. |
Independent monitoring of blood pressure by the patient can be essential in several cases. After serious cardiac events such as a heart attack, blood pressure needs to be monitored, as it can provide information about important risk factors such as hypertension. Uncontrolled blood pressure can contribute to a repeat heart attack or heart failure. When treating with blood pressure-lowering medications, such as ACE inhibitors, regular monitoring is crucial to avoid both over- and under-treatment, which can lead to kidney damage or circulatory problems. Studies indicate that a systolic blood pressure below 120 mmHg in patients with chronic kidney disease is associated with lower mortality, fewer severe cardiovascular events, and the preservation of cognitive functions.
Typically, blood pressure is measured using a non-invasive method, usually by inflating a cuff on the upper arm. The cuff is inflated above the systolic blood pressure, completely occluding the artery. As the cuff pressure is slowly released and falls below the systolic pressure, the heart begins to pump blood into the partially compressed artery. The pumping of the blood causes minimal pressure fluctuations within the cuff. The amplitude of these pressure fluctuations can then be used to determine the systolic and diastolic blood pressure values through various methods.
Another non-invasive method is measuring volume changes in the vascular system via an optical interface (photoplethysmography). Currently, these optical methods are not reimbursable under the statutory health insurance (GKV) and therefore are not covered under § 374a SGB V, and thus are not considered here.
For the initial specification (version 1) of HDDT, one mandatory interoperable value (MIV) is defined for blood pressure monitoring:
The table below lists typical use cases and certification relevant systems for this MIV:
| MIV | examples of typical use cases | examples of certification relevant systems |
|---|---|---|
| Blood Pressure Monitoring | • Preeclampsia prevention in pregnancies • Recording of blood pressure before a doctor’s visit • Monitoring blood pressure alongside medication |
• blood pressure cuff |
The MIV Blood Pressure Value covers values from measurements using a blood pressure cuff at home
| Defining ValueSet | The MIV Blood Pressure Value is defined by the FHIR ValueSet Blood Pressure Value from LOINC. This ValueSet contains LOINC codes for blood pressure measurements using a blood pressure cuff. The cuff may be placed at the upper arm or above the wrist. Different LOINC codes indicate whether blood pressure was measured using an oscillometric method, an auscultatory method, or a combination of both methods. |
| SMART Scopes | patient/Observation.rs?code:in=https://gematik.de/fhir/hddt/ValueSet/hddt-miv-blood-pressure-value patient/Device.rs patient/DeviceMetric.rs |
| FHIR Observation Profile | Device Data Recorders provide Blood Pressure Values to DiGA using the MIV-specific Observation Profile HDDT Blood Pressure Value. This profile allows to capture systolic, diastolic and mean arterial blood pressure values as a single FHIR Observation resource. |
| FHIR Interactions | Access to the resources is given through standard FHIR read and search RESTful interactions as described in the MIV-specific API. |
| Aggregated Data | By now there are no aggregated data defined for the MIV “Blood Pressure Value”. |
| Vendor Holdup | The maximum acceptable delay between data availability in the Health Record and data availability through the HDDT interface is 60 seconds. |
Historic-Data-Period |
Device Data Recorders that provide Blood Pressure Values MUST make the measured values retrievable for at least 30 days. |
Grace-Period |
A Device Data Recorder MAY reject a DiGA’s request for a patient’s Blood Pressure Values if the previous request for that patient was answered less than 5 minutes ago. |